
One of the most commonly used theoretical models is the International Association for the Properties of Water and Steam (IAPWS) formulation, which provides a reliable and accurate estimate of the density of water over a wide range of temperatures and pressures. The density of water at 15☌ can be calculated using experimental data or theoretical models. How is the density of water at 15☌ calculated? At standard atmospheric pressure (1 atm), the density of water increases by about 0.14% for every 100 atmospheres of pressure increase. The effect of pressure on the density of water is relatively small compared to the effect of temperature. As the pressure increases, the density of water increases, and as the pressure decreases, the density decreases. The density of water decreases as the temperature increases from 15☌ to boiling point (100☌), where the density is 958.4 kg/m³.Īnother factor that affects the density of water is pressure. As the temperature increases or decreases from 4☌, the density of water decreases.Īt 15☌, the density of water is lower than at 4☌, but it is still relatively high at 999.1 kg/m³. At this temperature, water reaches its maximum density, which means that a given volume of water contains the maximum amount of mass. At standard atmospheric pressure (1 atm), the density of water is highest at 4☌, where it is 1000 kg/m³. The density of water is affected by several factors, including temperature and pressure. 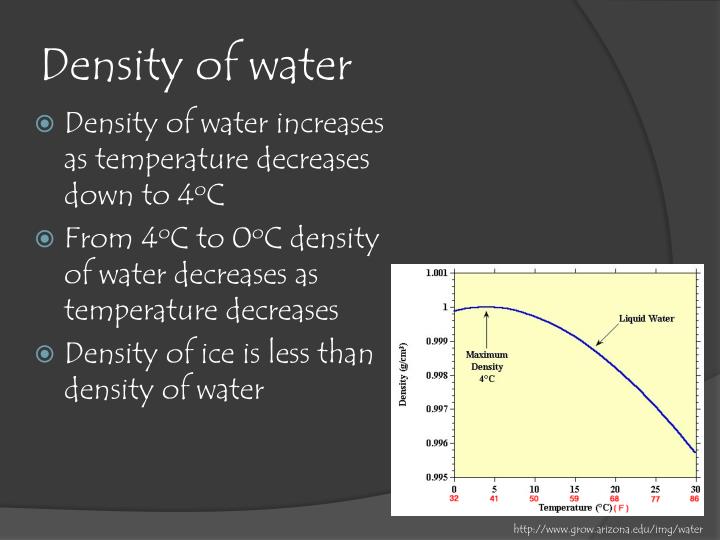
For example, the density of a small amount of water and the density of a large amount of water at the same temperature and pressure will be the same. Density is an intensive property, which means it does not depend on the size of the sample. 
Where mass is the amount of matter in an object, and volume is the amount of space that object occupies. It is expressed in units of mass per unit volume, such as kilograms per cubic meter (kg/m³) or grams per milliliter (g/mL). What is density?ĭensity is a measure of how much mass is contained in a given volume. At 15☌, the density of water is 997.05 kg/m³, which is slightly lower than the density at 4☌. Note that the density of water decreases as the temperature increases from 0☌ to 100☌, and the maximum density of water is reached at 4☌. The density of water at 15☌ is an important physical property that affects the behavior of water in various applications in science, technology, and environmental monitoring. The density of water decreases as the temperature increases or decreases from 15☌. The density of water at 15☌ is 999.1 kg/m³, which is slightly lower than the maximum density of water at 4☌. 
What is the density of water at 15 degrees Celsius? We will explore what density is and how it is calculated, what factors affect the density of water, and how density is related to other properties of water. In this blog post, we will focus on the density of water at a specific temperature, 15☌. The density of water, which is the measure of how much mass is contained in a given volume, is affected by temperature and pressure. It exists in three states, solid, liquid, and gas, depending on the temperature and pressure conditions. It covers approximately 71% of the earth’s surface and is a crucial component of living organisms. Water is an essential substance for life on earth.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
